Tyrosine kinase targeting uncovers oncogenic pathway plasticity in Tasmanian devil transmissible cancers
Tyrosine kinase targeting uncovers oncogenic pathway plasticity in Tasmanian devil transmissible cancers
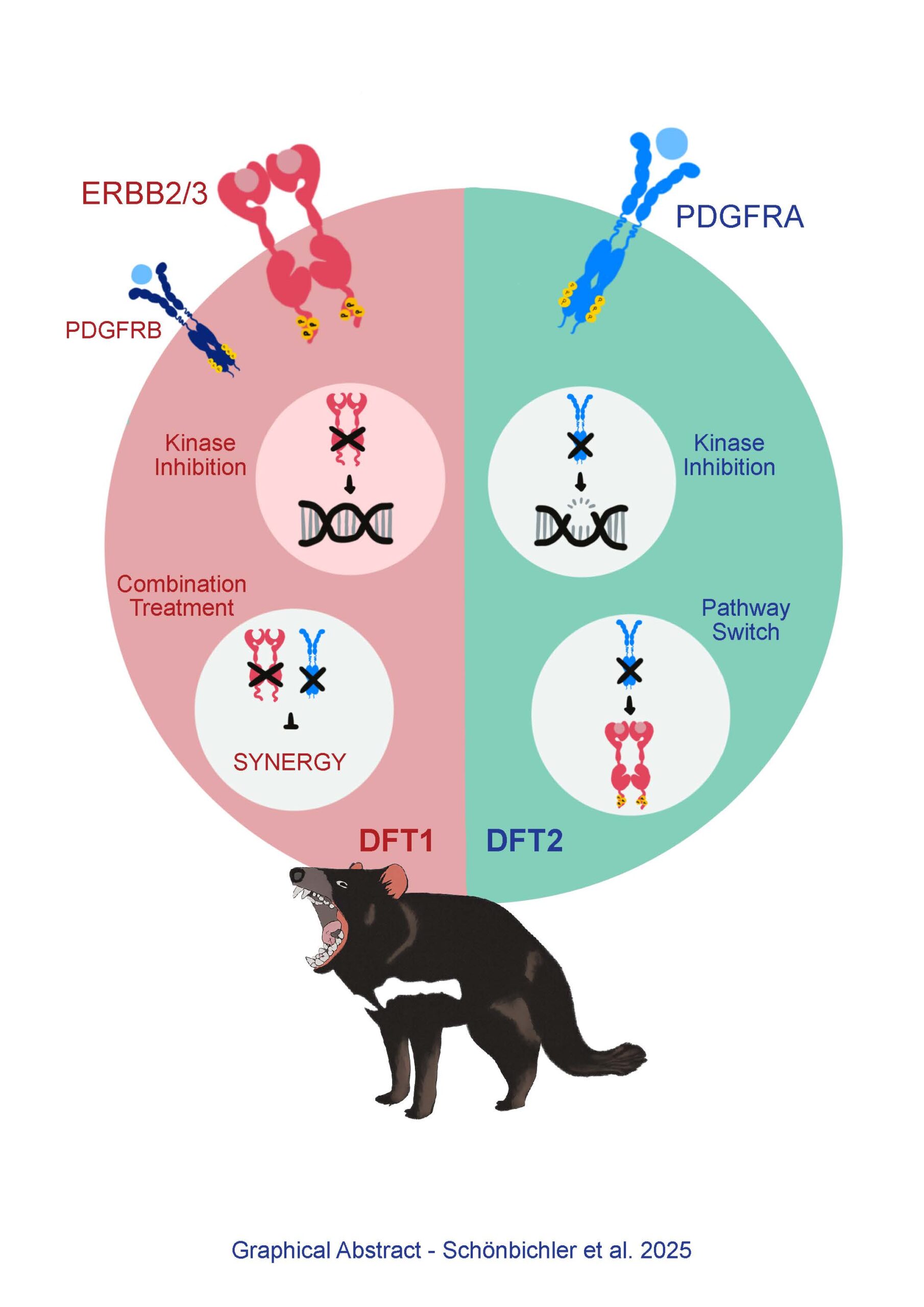
Two transmissible cancers, Devil Facial Tumour 1 (DFT1) and Devil Facial Tumour 2 (DFT2), have caused a significant decline in the Tasmanian devil population. DFT1 is driven by ERBB, while DFT2 is driven by PDGFRA, both growth factor receptor tyrosine kinases. We show that DFT cancer cells exhibit distinct kinase phosphorylation profiles that dictate their responses to tyrosine kinase inhibitors. Upon long-term treatment, both DFT cell lines develop resistance, with DFT1 cells rapidly evading ERBB inhibition without major DNA copy number alterations or significant changes in phosphorylation, suggesting signalling plasticity and engagement of alternative oncogenic drivers. In contrast, DFT2 cells exhibit a slowed development of resistance to imatinib, a selective tyrosine kinase inhibitor with known activity against PDGFRs. Moreover, DFT2 cell resistance is accompanied by DNA copy number alterations and an activation of ERBB and JAK/STAT signalling with MHCI downregulation, resembling DFT1 signalling. Dual targeting of ERBB and PDGFR shows synergistic effects in DFT1 and may prevent resistance emergence. These findings provide critical insight into the adaptive capacity of transmissible cancers and inform conservation strategies. Moreover, they highlight broader principles of kinase-driven resistance relevant to human cancers with high pathway plasticity.
The publication is now online.