Research projects
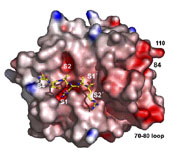
Cellular Pass Control: Antigen Processing and Presentation
The immune system recognizes both external invaders (bacteria and viruses) and internal aberrations (tumours). To this end, each cell has to identify itself towards the immune system by presenting intracellular peptides at the cell surface. Despite its enormous efficiency, some harmful cells escape the screening (pass control) of the immune system by presenting unsuspicious peptides only. We investigate the complex protease machinery involved in peptide processing. A detailed understanding promises treatment options against tumours and infectious diseases.
Undercover Agents – how pathogens escape cellular defence mechanisms
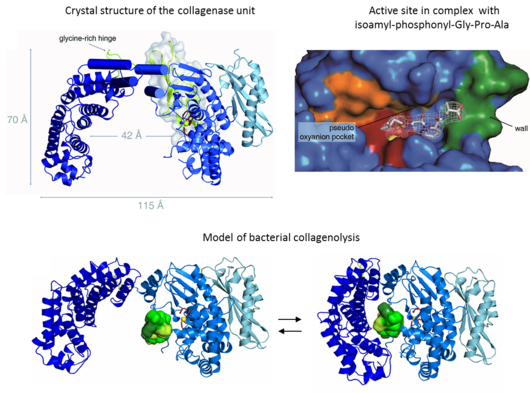
To undermine the defence mechanisms of the host, pathogens follow a classical strategy by imitating proteases of the host immune system. Of particular interest are bacterial collagenases which contribute to the host colonization and infiltration and thus enable host infection.
Structure of collagenase G reveals a chew-and-digest mechanism of bacterial collagenolysis
Eckhard U, Schönauer E, Nüss D & Brandstetter H (2011), Nat. Struct. Mol. Biol. 18: 1109–14
allergies
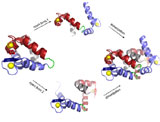
Allergens are foreign proteins that are per se harmless, but cause excessive immune reactions with approximately 20 % of the population, ranging from itching to anaphylactic shock. What biochemical properties render a protein allergenic? Employing structural investigations we could develop the molecular metamorphosis model that explains why (a family of) allergens elicit an allergic immune response: Triggered by intramolecular rearrangements, allergens undergo changes in oligomerization that will influence key events such as endocytosis, proteolytic processing and antigen presentation as well as antibody recognition and crosslinking. Suppression of the metamorphosis offers an innovative and highly specific therapeutic approach.
blood coagulation
The blood coagulation system stops life-threatening bleeding and at the same time prohibits similarly fatal vessel occlusion (thrombosis). To control the underlying reactions, nature employs a molecular proofreading principle, allowing blood coagulation only when different stimuli are simultaneously present. Failures in the fine-tuning of coagulation reactions imply bleeding disorders, e.g. haemophilia. At the heart of the blood coagulation regulation is factor IX where we could detect long-ranged intra- and intermolecular communication lines. These findings rationalize strategies for the treatment of bleeding disorders.
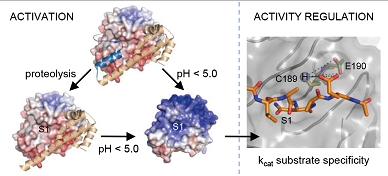
prostatic and epidermal kallikreins
Several tissue kallikreins (KLKs) are synthesised in the prostate, including KLK2, 3, 4 und 11. These serine proteases are interconnected by an activation web. Occurence of prostate cancer is accompanied by an exceeding increase of KLK3, aka PSA. Therefore, the PSA level serves as a diagnostic marker of prostate cancer, albeit prone to false positive signals. We expect to improve the diagnostic reliability by correlating the amount and the activity of all prostatic KLKs. Epidermal KLKs (KLK5, 7, 14) together with their regulator (LEKTI) play an important role in epidermal regeneration. Disturbance of the KLK regulation generates atopic dermatitis, because a first defence barrier gets disrupted.