Molecular Immunology
The Molecular Immunology Lab investigates molecular mechanisms of signaling processes and feedback regulation in immune pathologies, with a strong focus on signaling pathways in primary human immune cells. In earlier years, our research centered on the JAK/STAT pathway, where we identified STAT molecules as key contributors to the expression of type 2 inflammation-related genes. We also uncovered novel roles for SOCS proteins beyond their classical function in JAK/STAT regulation, including their involvement in signaling pathways triggered by pattern recognition receptors.
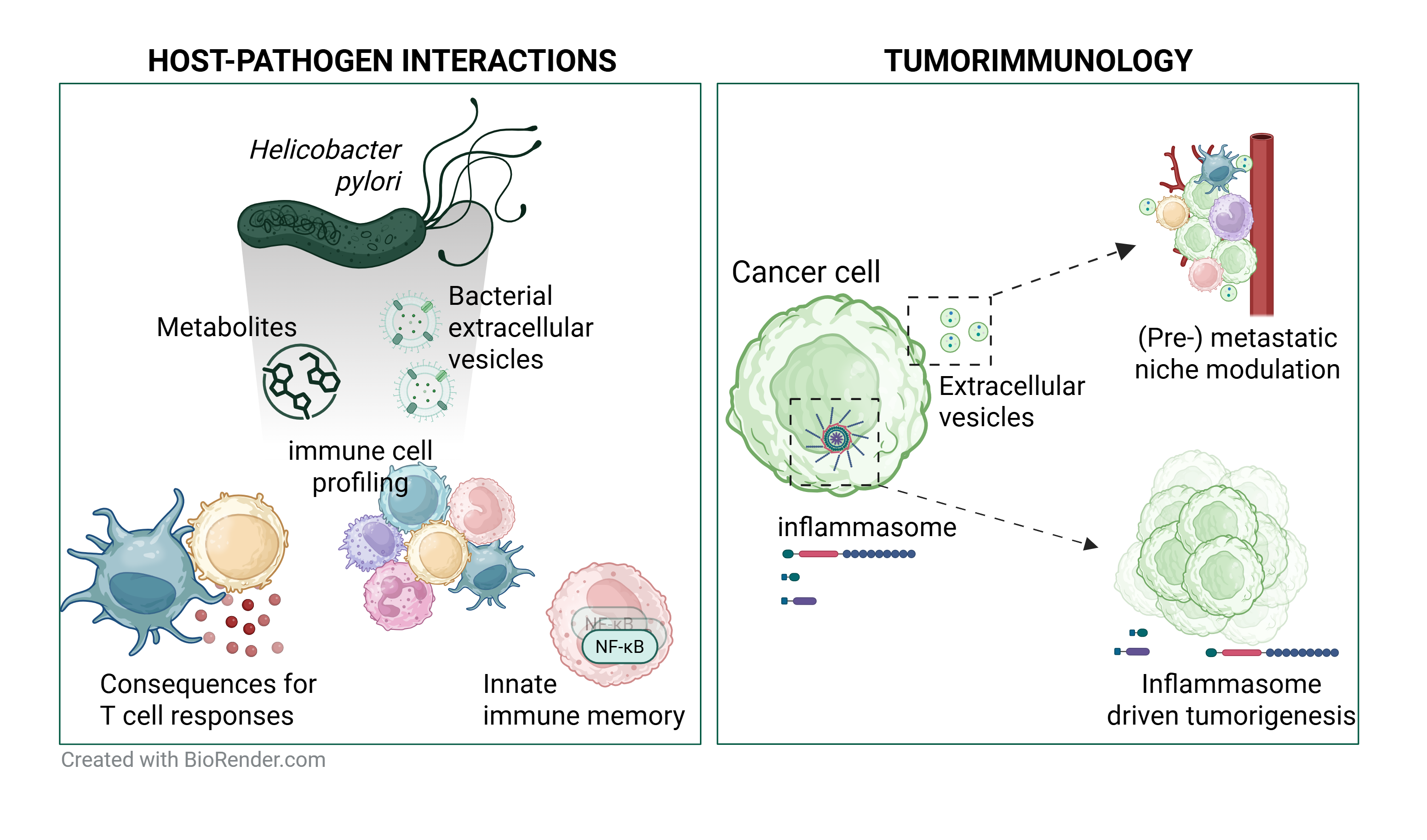
More recently, our research has shifted toward understanding the immunological impact of Helicobacter pylori infection and its role in chronic inflammation. We have gained valuable insights into the pathogen’s sophisticated strategies for immune evasion and persistence, identifying distinct forms of innate immune memory that are highly specific to Helicobacter pylori. These findings shed light on how the bacterium sustains chronic gastric inflammation and may drive the progression toward gastric cancer. Our group is also deeply interested in the role of NLRP3 and other inflammasome components as key players in inflammation within the myeloid compartment.
In our ongoing research projects, we investigate how the immune system responds to infectious and malignant threats, focusing on chronic inflammatory processes and their underlying cellular and molecular mechanisms.
- Host-pathogen interactions between innate immune cells and Helicobacter pylori (H. pylori).
- Inflammasome-mediated pathogenesis in the development and progression of acute myeloid leukemia (AML).
- Immunomodulatory functions of bacterial and mammalian extracellular vesicles (EVs) in the context of infections and metastasis.