Research Projects
Cellular Pass Control: Antigen Processing and Presentation
The immune system recognizes both external invaders (bacteria and viruses) and internal aberrations (tumours). To this end, each cell has to identify itself towards the immune system by presenting intracellular peptides at the cell surface. Despite its enormous efficiency, some harmful cells escape the screening (pass control) of the immune system by presenting unsuspicious peptides only. We investigate the complex protease machinery involved in peptide processing. A detailed understanding promises treatment options against tumours and infectious diseases.
Given the increasing number of requests for high quality legumain, we teamed up with Jena Bioscience to rapidly serve the community, https://www.jenabioscience.com/proteins/recombinant-proteins/protein-kinases-phosphatases-and-peptidases/peptidases/pr-967-legumain.
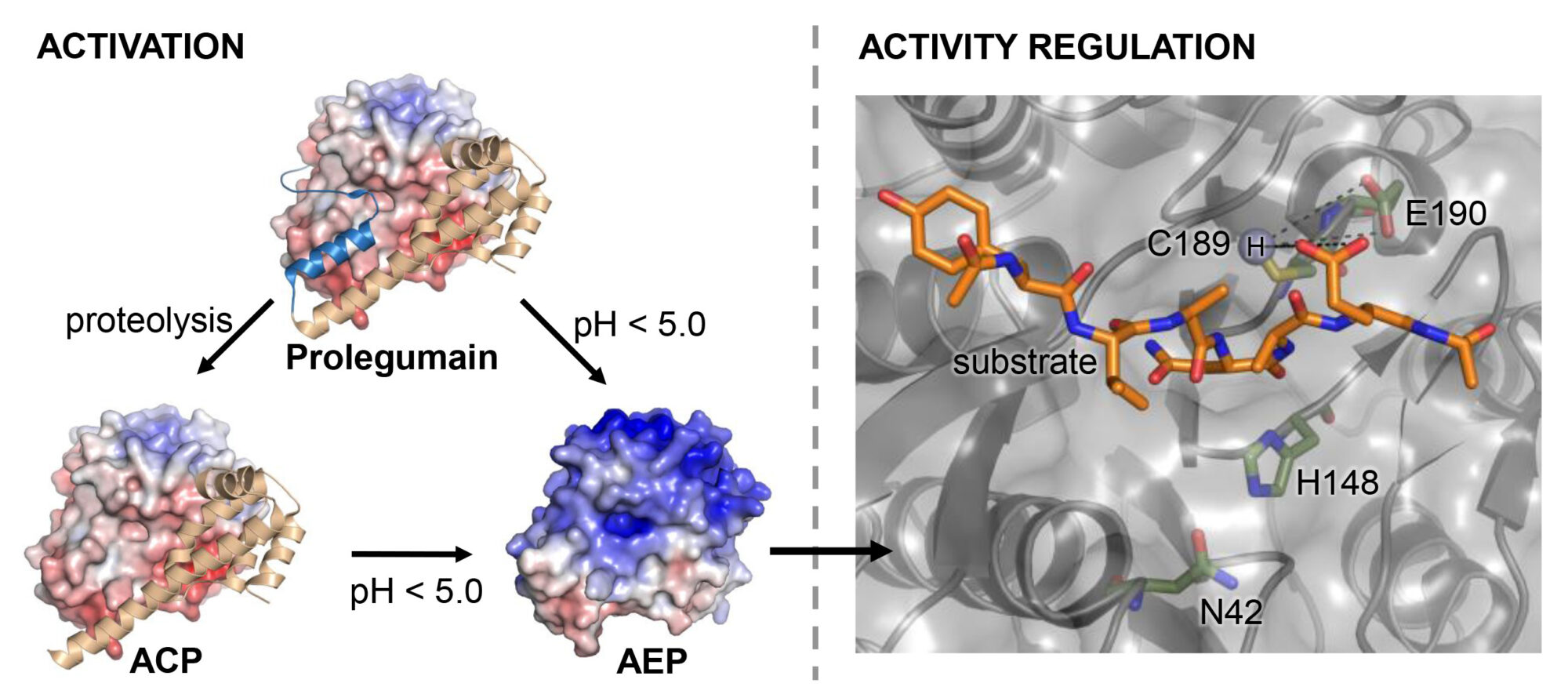
Elfriede Dall received an FWF Start Grant on „Functional Studies on Extra-lysosomal Legumain”. The project page can be found here.
Bacterial warfare – how bacterial collagenases as undercover agents evade the defences of the innate immune system
The extracellular matrix (ECM) constitutes one of the first critical physical defence line of the innate immune system against infections in humans and other mammals. Collagens form the resilient backbone of the mammalian ECM and ensure the integrity of all our tissues. For tissue invasion and host colonization, some human pathogens such as Hathewaya histolytica, Clostridium botulinum, Vibrio vulnificus or Leptospira interrogans specifically secrete bacterial collagenases to the collagenous scaffold of the ECM.
We target these bacterial collagenases in our research. We want to understand how they process the complex substrate collagen in detail, allowing us to develop highly inhibitors towards pathogen-secreted collagenases, on the one hand, and, on the other, to engineer custom-tailored collagenases for industrial and medical applications. This project was generously funded by the Austrian Science Fund (FWF) (P31843) and is headed by Dr. Esther Schönauer.
Learn more about our research on bacterial collagenases!
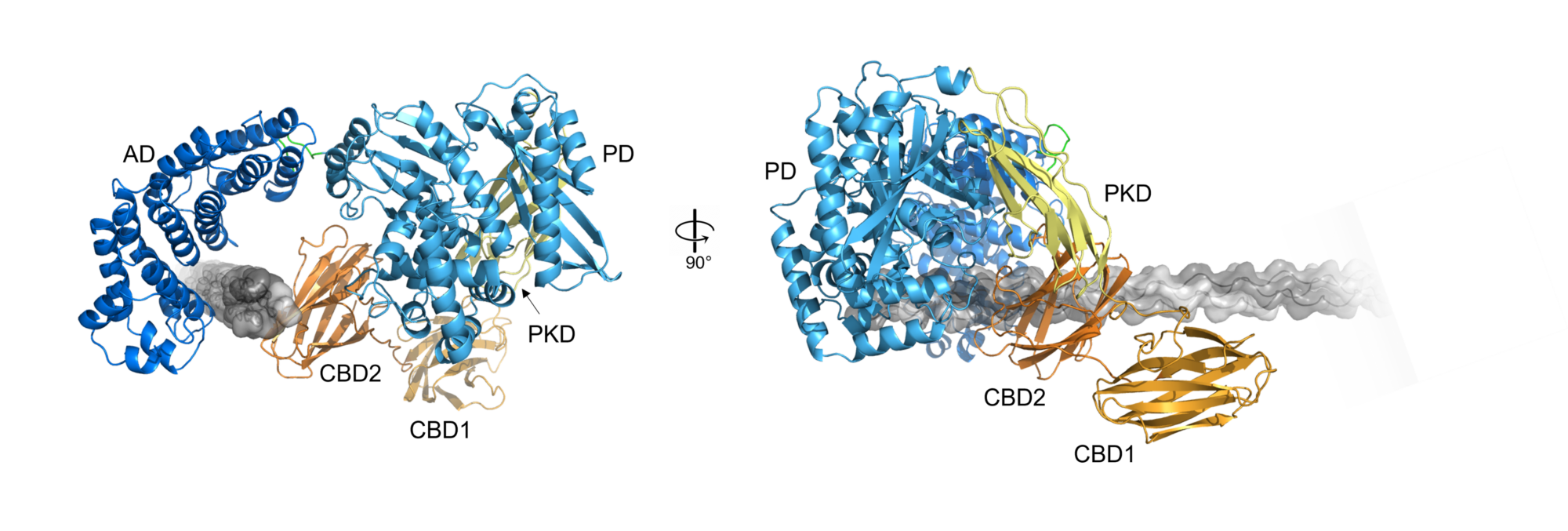
Structure and function of proprotein convertases
The serine protease furin has been connected to several pathologies such as rheumatoid arthritis, cancer and infectious diseases. In addition, many viruses require the proteolytic activation of their surface proteins by furin, including the measles virus, the denque virus, highly pathogenic avian influenza viruses and SARS-CoV-2. Thus, furin inhibitors are promising therapeutics against many diseases. The SARS-CoV-2 pandemic as well as the climate change related spread of tropical diseases, like denque fever, demonstrate the need for effective antiviral drugs.
Our work aims at a better understanding of the structure and fuction of proprotein convertases and structure-based development of novel furin inhibitors for the treatment of infections. Recently, a stand alone project (P36648-B) was granted to Dr. Sven O. Dahms by the Austrian Science Fund (FWF) to support this work.
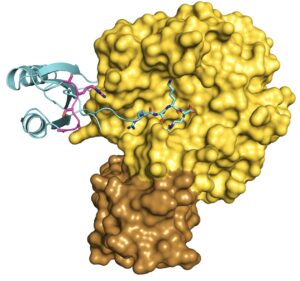
Figure: X-ray structure of furin (surface representation) in complex with the PC1-prodomain (cartoon representation). PDB-ID: 9FIE
Learn more about our research on proprotein-convertases.